Tykhon Zubkov
Associate Professor of Chemistry
About Me
I have been working at the Chemistry Department of Ball State University since 2007. My research interests involve understanding the growth of metal and semiconductor nanostructures and understanding how they enable catalytic and photocatalytic reactions on their surfaces. I got interested in Chemistry since middle school, thanks to a friend. Having a wonderful Chemistry teacher cemented this interest.
My College Experience
I received my Bachelor’s degree in Chemistry from Kyiv University, Ukraine, with the concentration in Physical Chemistry. My undergraduate research was in the area of heterogeneous catalysis. It brought me to the field of surface science and surface analysis, in which I focused in the Ph.D. program at University of Pittsburgh. This and my postdoctoral experience ultimately led me to become an analytical chemist.
What I have Learned?
After working in higher education and in a government lab and from talking to my friends who work in industry, I see how important it is for a chemist to have a solid basic understanding in all main branches of Chemistry. Many chemists morph from one branch to another throughout their careers. We should also be able to communicate chemical information to non-chemists because working in interdisciplinary teams is the new norm for many chemists.
Degree History
Postdoctoral Fellow Pacific Northwest National Laboratory (2007)
University of Pittsburgh Ph.D. (2004)
Kiev University Science Candidate (1997)
Kiev University B.S. (1993)
Research interests
Our general interests are in the chemistry of solid surfaces.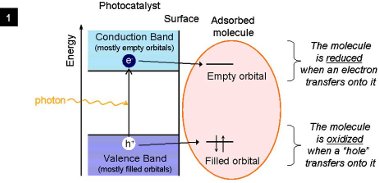
Specific Goal: Exploring new photocatalytic materials for oxidation of organic matter under UV or sunlight. These materials can have environmentally important applications such as decontamination, more economical waste treatment (including toxic waste), air purification, bactericidal and self-cleaning surfaces.
 The focus materials are transition metal sulfides and oxides, for example MoS2, WS2, TiO2. When irradiated by light, a photocatalyst can remove electrons from adsorbed molecules or it can inject extra electrons onto them, thus initiating further chemical reactions (see Figs. 1 and 2).
The focus materials are transition metal sulfides and oxides, for example MoS2, WS2, TiO2. When irradiated by light, a photocatalyst can remove electrons from adsorbed molecules or it can inject extra electrons onto them, thus initiating further chemical reactions (see Figs. 1 and 2).
In our research, we:

- Synthesize prospective photocatalysts if necessary.
- Characterize them by imaging and spectroscopic techniques, such as Transmission Electron Microscopy (TEM) (see Fig. 3), ultraviolet-visible absorption spectroscopy (UV vis), infrared spectroscopy (IR).
- Study how organic molecules adsorb on prospective photocatalysts.
- Analyze the obtained surface species under clean high-vacuum conditions.
- Monitor their chemical transformations induced by visible or ultraviolet light or by temperature. Transmission IR spectroscopy is the main analytical technique for identifying surface species and monitoring their reactions. IR- and UV-compatible vacuum cell is being constructed, in which IR spectroscopy of adsorbed species can be performed simultaneously with UV irradiation.
- Test the photocatalysts performance in the liquid or gas media. This is done using UV vis spectroscopy, High Performance Liquid Chromatography (HPLC), and in future, Gas Chromatography (GC or GC-MS combination).